After our first annual Pain Fellow’s Day and in anticipation of the Texas Pain Society 17th Annual Scientific Conference, we gathered information about fellowship programs around the state.
The University of Texas Health Science Center at Houston (UTHealth Houston) had one fellow, Karla Samaniego, in the McGovern Medical School's Department of Anesthesiology, Critical Care and Pain Medicine’s Pain Management Fellowship.
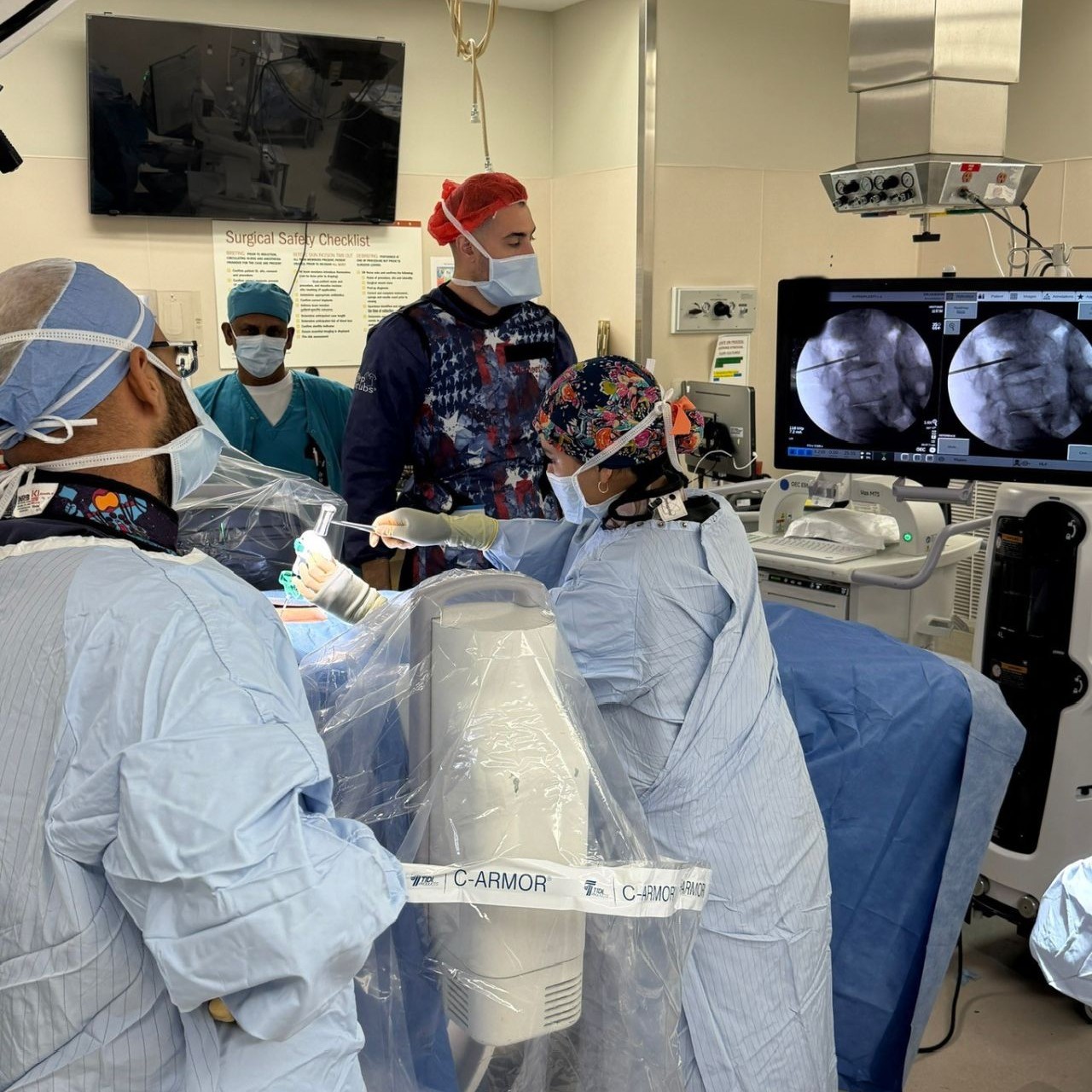 1. What makes this program unique for pain fellows?
1. What makes this program unique for pain fellows?
The program stands out for its high procedural and advanced procedural volume, giving fellows extensive exposure to everything from routine injections to cutting-edge techniques like spinal cord stimulation, intrathecal pumps, kyphoplasty, sacroiliac joint fusion, and peripheral nerve stimulation. It also provides flexibility to tailor the fellowship through elective rotations, including opportunities with private practice physicians, so fellows gain insight into different practice models. Ultimately, the focus is on ensuring fellows graduate with the skills and experience needed to thrive in private practice or academic settings, with strong procedural competence and a clear understanding of comprehensive pain management.



 George Polson MD
George Polson MD Grayson Koval, MD
Grayson Koval, MD Adedeji Olusanya DO, MPH
Adedeji Olusanya DO, MPH Vishal Bansal, MD
Vishal Bansal, MD  Joseph Liao, M.D.
Joseph Liao, M.D.
 Did you know that TPS has it's very own
Did you know that TPS has it's very own 







